Microwave Technology and Continuous-Flow Production






October 16, 2007
BY T. Michael Barnard and Nicholas E. Leadbeater
The breadth of biodiesel production technologies has exploded along with the expansive growth of the industry in the past several years. The biodiesel process continues to evolve as producers and researchers determine ways to refine production techniques.
Occasionally, drastic design changes provide a new way of thinking about the transesterification process. As the process evolves there are opportunities for inventive technology. Researchers in Connecticut have found and developed an innovative process they hope will move from the table top to the field.
In 1954, microwave ovens were introduced into homes, and their popularity has continued to grow. Simply put, when something is put into a microwave oven, it heats faster than when using conventional methods. Although microwave technology is widely used in homes, it has many other uses outside of the home. In fact, in industrial applications, microwaves are used for product drying, moisture and fat analysis of food products, solvent extraction applications and to irradiate coal to remove sulfur and other pollutants.
In the mid-1980s, home microwave ovens began being used for the production of chemicals in an organic chemistry laboratory setting. However, the home models weren't reliable or rugged enough to withstand the rigors of laboratory use. More importantly, they were unsafe for use with chemicals due to the possible build up of flammable vapors. These issues were addressed in the late 1990s with the introduction of microwave equipment specifically designed for use in laboratories.
Conventional thermal heating relies on conduction currents within the sample to provide an overall rise in temperature. Meanwhile, microwaves transfer energy directly into the sample. This thermal energy then drives the reaction to completion. By definition, this is a relatively slow process. Microwave irradiation is a form of energy that resides fairly low in the electromagnetic spectrum. It is slightly higher in frequency than radio waves, but much lower than X-rays, ultraviolet light and other chemical bond-breaking forms of energy. As a result, bonds are neither formed nor broken by microwave irradiation-it simply acts as an energy source, rapidly transferring energy to the sample.
Energy transferred directly into the sample, as accomplished by microwave irradiation, is powerful enough to accomplish chemical reactions in seconds or minutes, instead of the hours or days it would take using conventional thermal heating. For this reason, microwave irradiation has been accepted across a variety of scientific fields.
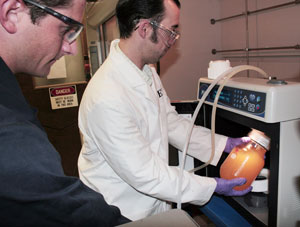
Realizing that microwave heating can speed up chemical reactions and improve the quality of the products formed, a research group at the University of Connecticut began using it for the production of biodiesel in 2006. The researchers demonstrated that, in a batch reactor, biodiesel could be made by heating oil, methanol and lye to 50 degrees Celsius (120 degrees Fahrenheit) in a microwave apparatus and maintaining this temperature for one minute.
Methanol is the most commonly used alcohol in the transesterification of vegetable oil to biodiesel. However, if replaced by ethanol it would be possible to obtain both starting materials from renewable resources. Ethanol is most commonly made from corn, but can also be produced from other feedstocks such as grain sorghum, wheat, barley or potatoes. The University of Connecticut researchers tested ethanol in the microwave apparatus. They found it was possible to convert vegetable oil to biodiesel with a quantitative conversion obtained after one minute at 50 degrees Celsius.
Although the initially developed process was fast, the test microwave units could only hold a maximum of three quarts per batch, limiting production to a small scale. However, continued efforts between the university and CEM Corp., a microwave apparatus manufacturer, have led to a continuous-flow approach for the preparation of biodiesel using microwave heating. The apparatus resulting from this collaboration can produce approximately two gallons of biodiesel per minute.
Energy consumption studies show that the continuous-flow preparation of biodiesel using microwave heating proves to be more energy efficient than the conventional (nonmicrowave) synthesis of biodiesel in large tank reactors. However, caution should be stressed when considering these results. There has been a report of the use of a domestic microwave for the reaction between methanol and seed oils in a batch process. However, production is limited due to the scale and the inherent reproducibility and safety issues affiliated with using a home microwave apparatus for making chemicals.
Three patent applications have been filed on the use of microwave heating in biofuels manufacture (WO 03/014272 A2, 2003; US 2005/0274065 A1, 2005; US 2006/0162245 A1, 2006). All three use a specially designed large microwave apparatus rather than readily available commercial scientific models.
The Microwave Accelerated Reaction System (MARS) used by the University of Connecticut and manufactured by CEM Corp. is a modular unit with a small footprint. One module could produce more than 800,000 gallons of biodiesel per year. Linking several units in parallel holds the potential for even greater production levels and a high-throughput design. This also offers redundancy. If the units are used in parallel and one fails, production can still continue at a slightly lower output. If a large tank reactor fails, production stops until repairs have been made. Also, because the microwave units are portable, it would be possible to take the microwave unit to the feedstock source rather than transport feedstock to a larger processing facility. For example, a cooperative of farms could own a unit and move it from site to site on an as-needed basis. One can also imagine the possibilities for fast-food restaurants. Since waste and virgin oils are common feedstocks, restaurants could convert fryer oil into biodiesel on site for use as a heating oil.
Microwave irradiation is revolutionizing many different industrial applications, and biodiesel production is merely the latest field to see the impact of this technology. Implementing multiple microwave units could have a major impact on land, energy and time savings compared with a single large reactor tank process. While microwave energy is still a relatively young technology in the biodiesel arena, it is a technology whose time has come.
T. Michael Barnard is a development chemist with CEM Corp. Reach him at mike.barnard@cem.com or (704) 821-7015. Nicholas E. Leadbeater is a professor at the University of Connecticut. Reach him at nicholas.leadbeater@uconn.edu or (860) 617-3518. This research was published in Energy and Fuels: 2006, Vol. 20, Issue 5 p. 2,281- 2,283–Batch; 2007, Vol. 21, Issue 3, p. 1,777-1,781–Continuous-Flow
The claims and statements made in this article belong exclusively to the author(s) and do not necessarily reflect the views of Biodiesel Magazine or its advertisers. All questions pertaining to this article should be directed to the author(s).
Occasionally, drastic design changes provide a new way of thinking about the transesterification process. As the process evolves there are opportunities for inventive technology. Researchers in Connecticut have found and developed an innovative process they hope will move from the table top to the field.
In 1954, microwave ovens were introduced into homes, and their popularity has continued to grow. Simply put, when something is put into a microwave oven, it heats faster than when using conventional methods. Although microwave technology is widely used in homes, it has many other uses outside of the home. In fact, in industrial applications, microwaves are used for product drying, moisture and fat analysis of food products, solvent extraction applications and to irradiate coal to remove sulfur and other pollutants.
In the mid-1980s, home microwave ovens began being used for the production of chemicals in an organic chemistry laboratory setting. However, the home models weren't reliable or rugged enough to withstand the rigors of laboratory use. More importantly, they were unsafe for use with chemicals due to the possible build up of flammable vapors. These issues were addressed in the late 1990s with the introduction of microwave equipment specifically designed for use in laboratories.
Conventional thermal heating relies on conduction currents within the sample to provide an overall rise in temperature. Meanwhile, microwaves transfer energy directly into the sample. This thermal energy then drives the reaction to completion. By definition, this is a relatively slow process. Microwave irradiation is a form of energy that resides fairly low in the electromagnetic spectrum. It is slightly higher in frequency than radio waves, but much lower than X-rays, ultraviolet light and other chemical bond-breaking forms of energy. As a result, bonds are neither formed nor broken by microwave irradiation-it simply acts as an energy source, rapidly transferring energy to the sample.
Energy transferred directly into the sample, as accomplished by microwave irradiation, is powerful enough to accomplish chemical reactions in seconds or minutes, instead of the hours or days it would take using conventional thermal heating. For this reason, microwave irradiation has been accepted across a variety of scientific fields.
Realizing that microwave heating can speed up chemical reactions and improve the quality of the products formed, a research group at the University of Connecticut began using it for the production of biodiesel in 2006. The researchers demonstrated that, in a batch reactor, biodiesel could be made by heating oil, methanol and lye to 50 degrees Celsius (120 degrees Fahrenheit) in a microwave apparatus and maintaining this temperature for one minute.
Methanol is the most commonly used alcohol in the transesterification of vegetable oil to biodiesel. However, if replaced by ethanol it would be possible to obtain both starting materials from renewable resources. Ethanol is most commonly made from corn, but can also be produced from other feedstocks such as grain sorghum, wheat, barley or potatoes. The University of Connecticut researchers tested ethanol in the microwave apparatus. They found it was possible to convert vegetable oil to biodiesel with a quantitative conversion obtained after one minute at 50 degrees Celsius.
Although the initially developed process was fast, the test microwave units could only hold a maximum of three quarts per batch, limiting production to a small scale. However, continued efforts between the university and CEM Corp., a microwave apparatus manufacturer, have led to a continuous-flow approach for the preparation of biodiesel using microwave heating. The apparatus resulting from this collaboration can produce approximately two gallons of biodiesel per minute.
Energy consumption studies show that the continuous-flow preparation of biodiesel using microwave heating proves to be more energy efficient than the conventional (nonmicrowave) synthesis of biodiesel in large tank reactors. However, caution should be stressed when considering these results. There has been a report of the use of a domestic microwave for the reaction between methanol and seed oils in a batch process. However, production is limited due to the scale and the inherent reproducibility and safety issues affiliated with using a home microwave apparatus for making chemicals.
Three patent applications have been filed on the use of microwave heating in biofuels manufacture (WO 03/014272 A2, 2003; US 2005/0274065 A1, 2005; US 2006/0162245 A1, 2006). All three use a specially designed large microwave apparatus rather than readily available commercial scientific models.
The Microwave Accelerated Reaction System (MARS) used by the University of Connecticut and manufactured by CEM Corp. is a modular unit with a small footprint. One module could produce more than 800,000 gallons of biodiesel per year. Linking several units in parallel holds the potential for even greater production levels and a high-throughput design. This also offers redundancy. If the units are used in parallel and one fails, production can still continue at a slightly lower output. If a large tank reactor fails, production stops until repairs have been made. Also, because the microwave units are portable, it would be possible to take the microwave unit to the feedstock source rather than transport feedstock to a larger processing facility. For example, a cooperative of farms could own a unit and move it from site to site on an as-needed basis. One can also imagine the possibilities for fast-food restaurants. Since waste and virgin oils are common feedstocks, restaurants could convert fryer oil into biodiesel on site for use as a heating oil.
Microwave irradiation is revolutionizing many different industrial applications, and biodiesel production is merely the latest field to see the impact of this technology. Implementing multiple microwave units could have a major impact on land, energy and time savings compared with a single large reactor tank process. While microwave energy is still a relatively young technology in the biodiesel arena, it is a technology whose time has come.
T. Michael Barnard is a development chemist with CEM Corp. Reach him at mike.barnard@cem.com or (704) 821-7015. Nicholas E. Leadbeater is a professor at the University of Connecticut. Reach him at nicholas.leadbeater@uconn.edu or (860) 617-3518. This research was published in Energy and Fuels: 2006, Vol. 20, Issue 5 p. 2,281- 2,283–Batch; 2007, Vol. 21, Issue 3, p. 1,777-1,781–Continuous-Flow
The claims and statements made in this article belong exclusively to the author(s) and do not necessarily reflect the views of Biodiesel Magazine or its advertisers. All questions pertaining to this article should be directed to the author(s).
Advertisement
Advertisement
Upcoming Events