The Chemical Kinetics of Glycerolysis


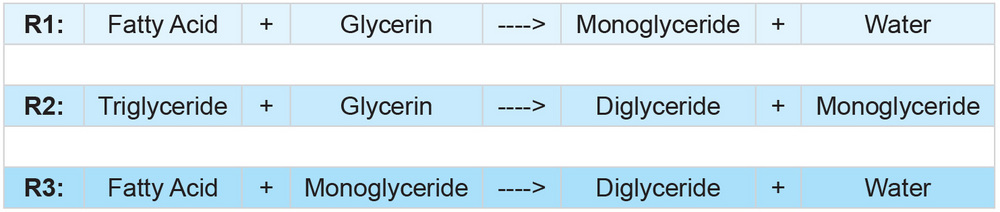
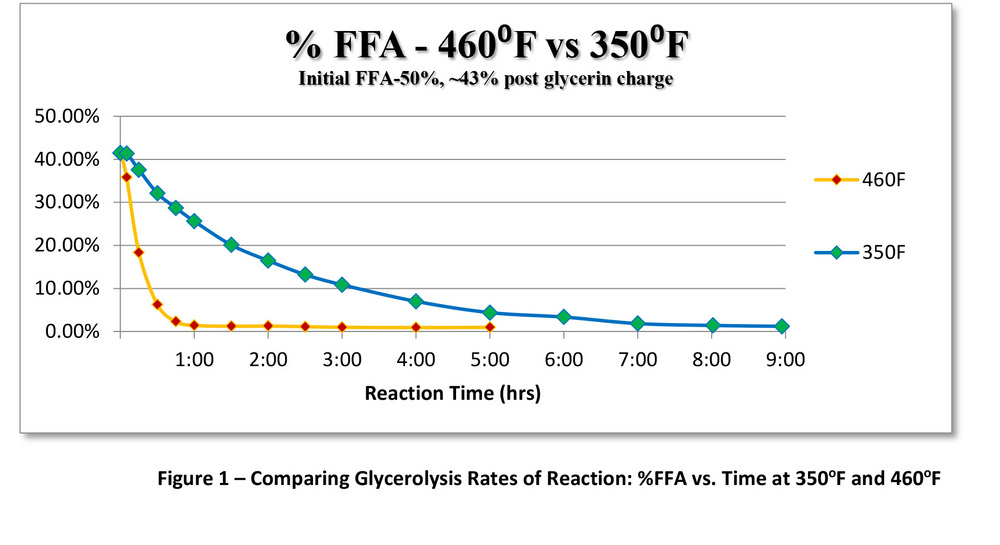


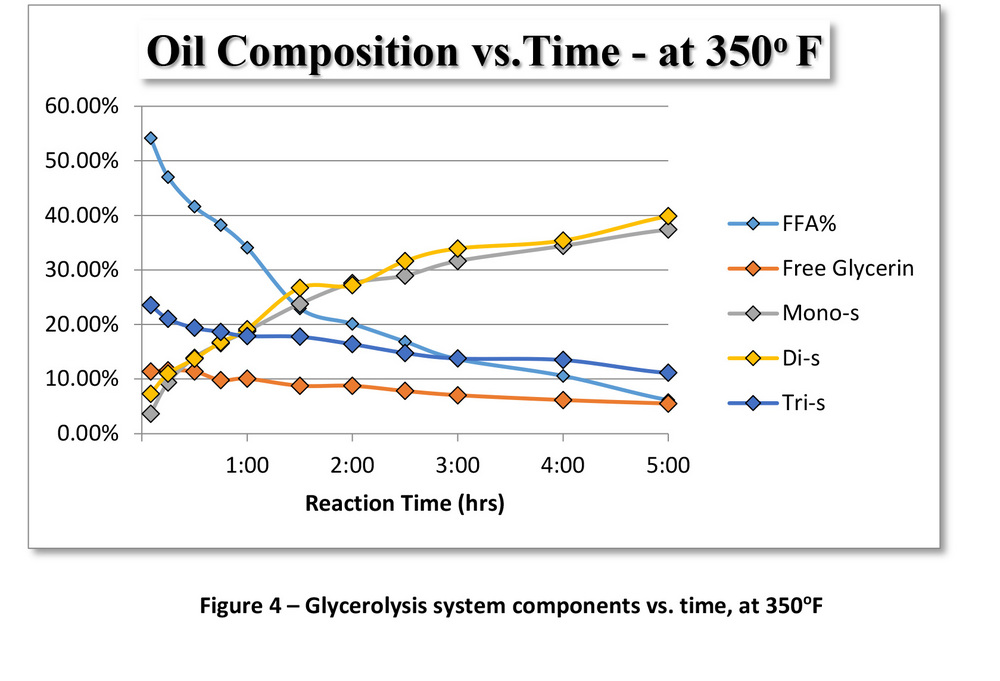
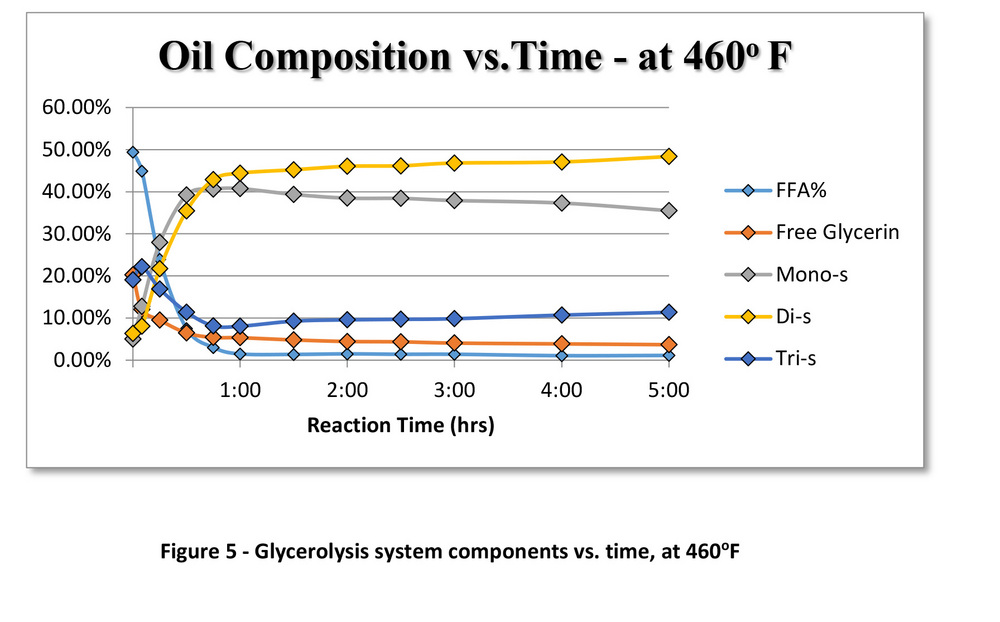

PHOTO: SUPERIOR PROCESS TECHNOLOGIES LLC
May 15, 2014
BY Erik Anderson
The question of what oil pretreatment method is the best is an ongoing conversation among biodiesel producers and engineers. Most low-grade feed oils contain high levels of free fatty acids (FFA), which can cause soap formation in traditional biodiesel processes. Several different pretreatment methods are used commercially to assimilate or remove FFA: acid esterification, vacuum steam stripping, caustic washing and glycerolysis; we consider enzymatics to be at the precommercial stage. The most prevalent pretreatment method has traditionally been acid esterification, since it can be done at relatively low process temperatures. The goal of acid esterification is the direct conversion of FFA into methyl esters (biodiesel) using sulfuric acid as a catalyst, with an excess of methanol. During acid esterification, each mole of fatty acid converted to methyl esters produces one mole of water. The resulting wet methanol must then be decanted, neutralized and dried via fractional distillation with high reflux rates, before it can be reused. Methanol drying columns can cost millions of dollars and are the biggest users of plant energy. By not having to dry wet methanol after acid esterification, biodiesel plants can cut their thermal energy consumption in half.
Alternatively, glycerolysis reduces the amount of FFA in low-grade oils without use of acid or methanol, and enables them to be converted into final product, rather than removing them and reducing product yield. The resulting glycerides formed during glycerolysis are then converted directly to biodiesel via base-catalyzed transesterification. Also, glycerolysis is done at high enough temperatures to completely dry the feed oil before the transesterification process, thus avoiding the formation of excess soaps and the decanting problems that can result.
Over the past decade, glycerolysis has continued to grow in popularity among those companies successful in the industry. For example, recent articles in several industry periodicals have noted that some biodiesel producers have been using glycerolysis successfully for several years (e.g., Renewable Energy Group’s Seneca, Ill., plant).
The rate of the glycerolysis reaction is determined by two variables: the initial concentration of FFA and temperature. Many biodiesel plants run their processes using steam heating systems, and are limited to operating temperatures of 350 degrees Fahrenheit or less. Although glycerolysis can be run at these lower temperatures, reaction kinetics are vastly improved when run at temperatures at or above 450 F. However, operating temperatures approaching 500 F are not recommended due to possible glycerin decomposition, forming acrolein.
Some biodiesel producers may not be familiar or comfortable with high-temperature processes, and therefore tend to shy away from the use of thermal oil heating systems needed for glycerolysis operating temperatures. This concern over the use of hot oil systems is due to a lack of industrial experience, particularly with oleochemicals.
Another benefit from glycerolysis is its simplicity. The only reagent needed for successful glycerolysis is glycerin, the byproduct of transesterification. In plants using glycerolysis, the glycerin produced during transesterification can be recycled back into the process, and the excess glycerin can be refined for sale as a valuable byproduct.
Advertisement
Research at Superior Process Technologies was done to compare glycerolysis at various operational temperatures. Multiple laboratory batch-wise glycerolysis reactions were performed on brown grease at 350 F and 460 F, representing steam-heated and thermal-oil-heated systems. The lab work and data analysis was performed by Chris Sorensen with SPT. Samples were taken over the course of the reaction and run on a Gas Chromatograph-Flame Ionization Detector to determine compositional makeup versus reaction time. The initial brown grease was determined to have an acid number of 100 (with 50 percent FFA) via wet chemistry titration prior to glycerolysis. Each batch was brought up to their respective temperatures under inert conditions using a nitrogen purge before charging glycerin. Eight batches were run in total at varying temperatures and FFA concentrations. By graphing the average acid numbers versus time of each reaction, the difference in the rate of FFA reduction can be seen in Figure 1.
At 460 F, the FFA concentration is lowered rapidly in the first hour, and well below 1 percent FFA within several hours. In comparison, the reaction at 350 F did lower the FFA below 2 percent, but required nine hours of reaction time. Clearly, the main cause for the observed slower reaction rate is the lower reaction temperature.
Another contributing factor that may affect reaction kinetics is the system’s ability to purge water out at 350 F compared to 460 F. Kirk Cobb, senior process engineer at SPT, points out that any residual mole fraction of water remaining in the reaction liquid may restrict the chemical kinetics at the end of the reaction. It was noted in the lab data that the 350 F batches could never quite reduce the remaining FFA below 1 percent, as the higher temperature batches were able to do. Any remaining water shifts the steady-state equilibrium slightly towards the reactants’ side of the equation, slowing the reaction. One solution to the problem of any remaining traces of water is to place the system under vacuum. At negative pressures, the water will be more effectively driven out, and an increase in the system kinetics can be achieved. The drawback to this approach is that there is a fine line between removing water and removing free glycerin, even at 350 F. Also, if vacuum leaks were to occur at glycerolysis temperatures, any air in the system can cause oxidation of the oil. An alternative is to run the reactions at atmospheric pressure, but use nitrogen to purge any water vapor. In large-scale, continuous glycerolysis operations, a combination of heat and nitrogen purging can be used with great success.
Combining glycerin with oils containing FFA, while heating, appears to result in three primary reactions shown in Figure 2.
The reaction of FFA and glycerin to form monoglycerides and water is initially the primary reaction (R1) during glycerolysis; this reaction is responsible for the majority of FFA reduction. A secondary reaction (R2) between glycerin and triglycerides forms mono- and diglycerides; one mole of triglyceride will react with one mole of free glycerin to form one mole each of mono- and diglyceride. A third reaction (R3) is the combination of FFA with monoglycerides to form diglycerides and water; this reaction may become dominant later in the system as monoglyceride concentrations increase, and free glycerin concentration diminishes with time.
The oil composition for FFA, free glycerin, monoglycerides, diglycerides and triglycerides was determined using GC-FID. This data was analyzed to determine the rate constant, K, for each of these three competing reactions, as shown in Figure 3.
Advertisement
The “rate constant” is a number that quantifies the speed of the reaction and is dependent only on temperature. The larger the K value, the faster that reaction. The actual laboratory data was modeled mathematically to determine the K values shown in Figure 3. Although the K value in each case is only a function of temperature, the total rate of reaction for each of these reactions is also dependent on the concentration of the reactants. Early in the batch reaction when FFA and free glycerin are relatively high, the R1 and R2 reactions will dominate; but as monoglyceride concentrations increase over time, the R3 reaction may begin to dominate the final consumption of FFA. All three of these reactions occur during high-temperature glycerolysis at different rates, and reach different states of equilibrium.
Figure 3 shows how the K values for glycerolysis at 350 F were significantly lower than the K values at 460 F.
One point of interest in Figure 3 is that, at 460 F, the rate constant of Reaction R3 (K=1.47) is almost as high as Reaction R1 (K=1.56). A possible explanation for the similar reaction rates observed is that monoglycerides, like free glycerin, have readily available alcohol groups that can react with FFA, with minimal steric hindrance. The results suggest that FFA does not discern much of a difference between bonding with glycerin or monoglycerides.
The K values for each of the reactions were determined using the compositional data at 350 F (Figure 4) and 460 F (Figure 5). Glycerolysis at 460 F reaches an equilibrium state within one to two hours, while the reaction at 350 F (steam temperatures) continues well beyond five hours. At 350 F, the composition of the oil follows the same trends as glycerolysis at 460 F, just at a slower rate. After a long enough timeline, both batch reactions might eventually produce the same results. The benefit of the higher-temperature glycerolysis becomes apparent when designing a full-scale biodiesel plant. In order to be able to transfer this process data from a batch system to a large-scale continuous system, understanding and accounting for the different reaction kinetics is essential. By increasing the rate of the reaction at the higher operating temperature, biodiesel manufacturers would be able to convert larger volumes of high-FFA oils to biodiesel at less expense.
Whether glycerolysis is performed at 350 or 460 F, the end products are essentially the same. Both contain high mole fractions of mono- and diglycerides along with lowered levels of FFA, and some remaining triglycerides and free glycerin. Glycerolysis at the high temperature has the added benefit of lowering FFA concentrations significantly faster. When scaled up to a continuous system, running glycerolysis at 460 F instead of 350 F will result in at least three times the amount of throughput, while producing an almost completely dry product. The end result is dry, glyceride-rich oil with FFA content below 1 percent. This study was conducted using very low-grade brown grease in order to be conservative on promoting the advantages of glycerolysis. In reality, this process works equally well when using a source of oil like yellow grease, with FFA levels ranging from 5 to 15 percent, although any level of FFA can be glycerized successfully.
The “chemical kinetics” results of this laboratory batch study have been successfully used to scale up the glycerolysis process to a continuous, industrial design. Readers of this article, who are interested in taking advantage of the glycerolysis process for biodiesel feed oil pretreatment, are encouraged to contact us.
Author: Erik Anderson
Laboratory Manager, Superior Process Technologies
612-378-0800 x5
eanderson@superiorpt.com
Related Stories
EcoCeres Inc., a leading pure-play renewable fuels producer, is partnering with Xiamen Airlines to collect and transport waste cooking oil from selected restaurant partners through its established supply chain.
The USDA maintained its outlook for 2025-’26 soybean oil use in biofuel production in its latest World Agricultural Supply and Demand Estimates report, released Aug. 12. The forecast for soybean oil prices was also unchanged.
U.S. soybean production for 2025 is forecast at 4.29 billion bushels, down 2% when compared to last year, according to the USDA National Agricultural Statistics Service’s latest monthly Crop Production report, released Aug. 12.
California’s new specified source feedstock attestation requirement: A critical new compliance step for renewable fuel producers
As of July 2025, California’s SCFS requires renewable fuel producers using specified source feedstocks to secure attestation letters reaching back to the point of origin. This marks a significant shift in compliance expectations.
At the University of Missouri, plant biochemist Jay Thelen is using arabidopsis as a powerful model to explore ways to boost oil production — an important step toward creating more sustainable, plant-based energy sources.
Upcoming Events